MMR Vaccine (Measles, Mumps and Rubella):
Is It Safer Than Measles?
 1. WHAT ARE SIDE EFFECTS OF THE MMR VACCINE?
1. WHAT ARE SIDE EFFECTS OF THE MMR VACCINE?
Common side effects of the MMR vaccine include fever, mild rash, and swelling of glands in the cheeks or neck.1 A more serious side effect is seizure, which occurs in about 1 in 640 children vaccinated with MMR2—about five times more often than seizure from measles infection.3

The World Health Organization (WHO) states that serious allergic reactions to the vaccine occur in about 1 in 100,000 doses.4 However, other severe side effects include deafness, long-term seizures, coma, lowered consciousness, permanent brain damage, and death.1 While the Centers for Disease Control and Prevention (CDC) states that these side effects are rare, the precise numbers are unknown.1 Additionally, the manufacturer’s package insert states, “M-M-R II has not been evaluated for carcinogenic or mutagenic potential, or potential to impair fertility.”5

 2. HOW ARE RISKS OF VACCINE SIDE EFFECTS MEASURED?
2. HOW ARE RISKS OF VACCINE SIDE EFFECTS MEASURED?
Methods to measure vaccine risks include surveillance systems, clinical studies, and epidemiological studies.
 3. HOW ACCURATE IS SURVEILLANCE OF ADVERSE EVENTS FROM THE MMR VACCINE?
3. HOW ACCURATE IS SURVEILLANCE OF ADVERSE EVENTS FROM THE MMR VACCINE?
The government tracks reported cases of vaccine side effects through the Vaccine Adverse Event Reporting System (VAERS). Approximately 40 cases of death and permanent injury from the MMR vaccine are reported to VAERS annually.6 However, VAERS is a passive reporting system—authorities do not actively search for cases and do not actively remind doctors and the public to report cases. These limitations can lead to significant underreporting.7 The CDC states, “VAERS receives reports for only a small fraction of actual adverse events.”8 Indeed, as few as 1% of serious side effects from medical products are reported to passive surveillance systems,9 and as few as 1.6% of MMR-related seizures are reported to VAERS.10 In addition, VAERS reports are not proof that a side effect occurred, as the system is not designed to thoroughly investigate all cases.11 As a result, VAERS does not provide an accurate count of MMR vaccine side effects.
 4. HOW ACCURATE ARE CLINICAL TRIALS OF THE MMR VACCINE?
4. HOW ACCURATE ARE CLINICAL TRIALS OF THE MMR VACCINE?
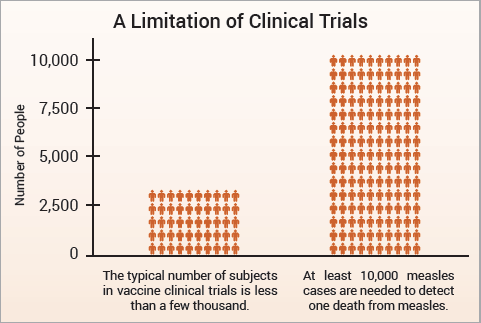
The CDC states, “Prelicensure trials are relatively small—usually limited to a few thousand subjects—and usually last no longer than a few years. Prelicensure trials usually do not have the ability to detect rare adverse events or adverse events with delayed onset.”7 Since measles is fatal in about 1 in 10,000 cases and results in permanent injury in about 1 in 80,000 cases,3 a few thousand subjects in clinical trials are not enough to prove that the MMR vaccine causes less death and permanent injury than measles (Fig. 1). In addition, the lack of adequate clinical trials of the MMR vaccine resulted in the manufacturer’s package insert data to be reliant on passive surveillance for rates of MMR-related neurological adverse reactions, permanent disability, and death.5
Figure 1: There are not enough subjects in clinical trials to prove that the measles vaccine poses less risk than measles.
 5. HOW ACCURATE ARE EPIDEMIOLOGICAL STUDIES OF THE MMR VACCINE?
5. HOW ACCURATE ARE EPIDEMIOLOGICAL STUDIES OF THE MMR VACCINE?
Epidemiological studies are hindered by the effects of chance and possible confounders—additional factors that could conceivably affect the groups being studied. For example, there is a well-known 2002 Danish study published in the New England Journal of Medicine involving about 537,000 children that looked for an association between the MMR vaccine and certain adverse events.12 The raw data in the study was adjusted, in an attempt to account for potential confounders, and the study found no association between the MMR vaccine and the adverse events. However, because there is no evidence that the estimated confounders used to adjust the raw data were actually confounders, the study did not rule out the possibility that the MMR vaccine increases the risk of an adverse event that leads to permanent injury by up to 77%. Consequently, the study did not rule out the possibility that such adverse events might occur up to four times more often than death from measles: 1 in 2,400 compared to 1 in 10,000 (Fig. 2 and Table 1). The range of possibilities found in the study, between the adjusted data and the raw data, makes the result inconclusive; even large epidemiological studies are not accurate enough to prove that the MMR vaccine causes less death or permanent injury than measles.
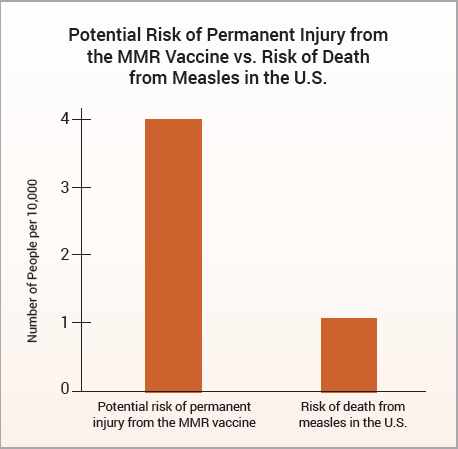
Figure 2: A 2002 Danish study did not rule out the possibility that the MMR vaccine can cause an adverse event leading to permanent injury four times more often than measles can be fatal.

 6. IS THE MMR VACCINE SAFER THAN MEASLES?
6. IS THE MMR VACCINE SAFER THAN MEASLES?
It has not been proven that the MMR vaccine is safer than measles. The vaccine package insert raises questions about safety testing for cancer, genetic mutations, and impaired fertility. Although VAERS tracks some adverse events, it is too inaccurate to measure against the risk of measles. Clinical trials do not have the ability to detect less common adverse reactions, and epidemiological studies are limited by the effects of chance and possible confounders. Safety studies of the MMR vaccine are particularly lacking in statistical power. A review of more than 60 MMR vaccine studies conducted for the Cochrane Library states, “The design and reporting of safety outcomes in MMR vaccine studies, both pre- and post-marketing, are largely inadequate.”13 Because permanent sequelae (aftereffects) from measles, especially in individuals with normal levels of vitamin A, are so rare,3 the level of accuracy of the research studies available is insufficient to prove that the vaccine causes less death or permanent injury than measles.
REFERENCES
- Centers for Disease Control and Prevention. Washington, D.C.: U.S. Department of Health and Human Services. Vaccines and immunizations: MMR vaccine side effects. [updated 2017 May 8; cited 2017 Jun 21]. https://www.cdc.gov/vaccines/vac-gen/side-effects.htm#mmr.
- Vestergaard M, Hviid A, Madsen KM, Wohlfahrt J, Thorsen P, Schendel D, Melbye M, Olsen J. MMR vaccination and febrile seizures: evaluation of susceptible subgroups and long-term prognosis. JAMA. 2004 Jul 21;292(3):356.
- Physicians for Informed Consent. Newport Beach (CA): Physicians for Informed Consent. Measles – disease information statement (DIS); updated 2019 Sep. https://www. physiciansforinformedconsent.org/measles/dis.
- World Health Organization. Measles vaccines: WHO position paper. Wkly Epidemiol Rec. 2009 Aug 28;84(35):355.
- Merck. Whitehouse Station (NJ): Merck and Co., Inc. M-M-R II (measles, mumps, and rubella virus vaccine live); revised 2017 May [cited 2019 Aug 4]. https://www.merck.com/product/usa/pi_circulars/m/mmr_ii/mmr_ii_pi.pdf.
- Centers for Disease Control and Prevention. Washington, D.C.: U.S. Department of Health and Human Services. CDC wonder: about the Vaccine Adverse Event Reporting System (VAERS);[cited 2017 Jun 21]. https://wonder.cdc.gov/vaers.html. Query for death and permanent disability involving all measles-containing vaccines, 2011-2015.
- Centers for Disease Control and Prevention. Manual for the surveillance of vaccine-preventable diseases. 5th ed. Miller ER, Haber P, Hibbs B, Broder K. Chapter 21: surveillance for adverse events following immunization using the Vaccine Adverse Event Reporting System (VAERS). Atlanta: Centers for Disease Control and Prevention; 2011. 1,2,8.
- Vaccine Adverse Event Reporting System. Washington, D.C.: U.S. Department of Health and Human Services. Guide to interpreting VAERS data; [cited 2017 Jun 21]. https://vaers.hhs.gov/data/dataguide.html.
- Kessler DA. Introducing MEDWatch. A new approach to reporting medication and device adverse effects and product problems. JAMA. 1993 Jun 2;269(21):2765-8
- Doshi P. The unofficial vaccine educators: are CDC funded non-profits sufficiently independent? [letter]. BMJ. 2017 Nov 7 [cited 2017 Nov 20];359:j5104. http://www.bmj.com/content/359/bmj. j5104/rr-13.
- Centers for Disease Control and Prevention. Washington, D.C.: U.S. Department of Health and Human Services. CDC wonder: about the Vaccine Adverse Event Reporting System (VAERS);[cited 2017 Jun 21]. https://wonder.cdc.gov/vaers.html.
- Madsen KM, Hviid A, Vestergaard M, Schendel D, WohlFahrt J, Thorsen P, Olsen J, Melbye M. A population-based study of measles, mumps, and rubella vaccination and autism. N Engl J Med. 2002 Nov 7;347(19):1477,1480.
- Demicheli V, Rivetti A, Debalini MG, Di Pietrantonj C. Vaccines for measles, mumps and rubella in children. Cochrane Database of Syst Rev. 2012 Feb 15;(2).
These statements are intended for informational purposes only and should not be construed as personal medical advice.
© 2019 Physicians for Informed Consent, an independent 501(c)(3) nonprofit educational organization. Updated Dec 2019.